|
|
Dr. Jiye Fang's Nano Research Group |
Single-atom catalysts (SACs) have emerged as a frontier in catalysis, offering exceptional performance and atomic efficiency. By dispersing individual metal atoms on suitable supports, SACs maximize the utilization of precious metals while providing unique opportunities to tailor catalytic activity and selectivity. This approach has shown strong potential for improving a wide range of important catalytic and electrocatalytic reactions, including small organic molecule oxidation and the oxygen reduction reaction (ORR) in fuel cells, the oxygen evolution reaction (OER) and hydrogen evolution reaction (HER) in water electrolysis, the electrochemical CO2 reduction reaction (eCO2RR), the water–gas shift (WGS) reaction (CO + H2O → CO2 + H2), and hydrogenation processes.
In supported SAC systems, isolated metal atoms are dispersed on or coordinated with surface atoms of appropriate support materials. These well-defined atomic sites not only maximize metal efficiency but also create unique electronic and geometric environments that can significantly enhance catalytic performance.
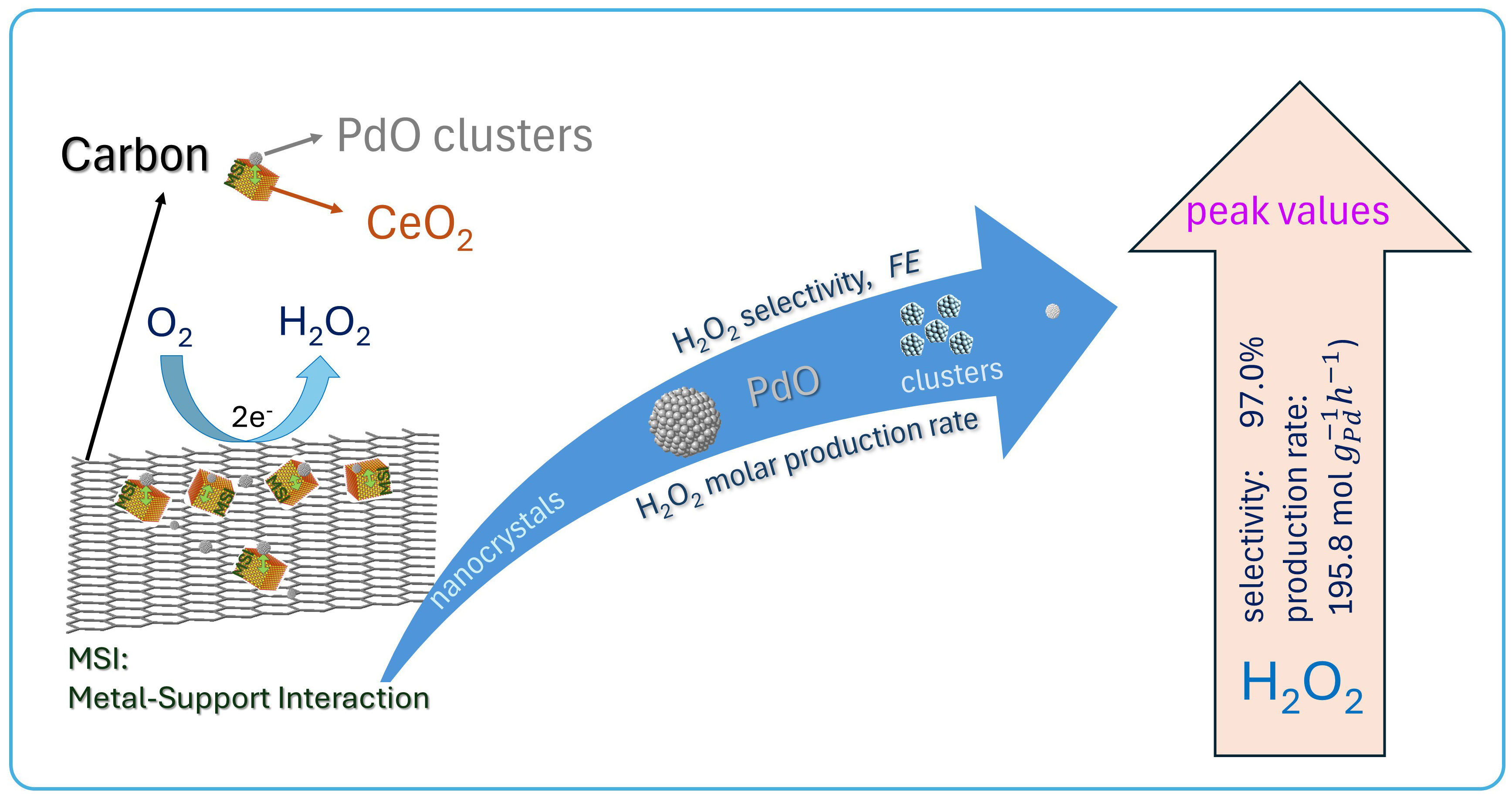
Our recent work focuses on palladium (Pd)-based single-atom catalysts and clusters for hydrogen peroxide (H2O2) electrosynthesis. This research is supported by the New York State Energy Research and Development Authority (NYSERDA). In this project, isolated reactive metal adatoms are deposited into host metal surfaces composed of relatively inert metals, enabling precise control over catalytically active sites. The catalytic performance of these materials strongly depends on the composition, structure, and metal–support interactions within the catalyst system. In particular, we are exploring PdO–CeO2–carbon catalyst systems, where strong metal–support interactions can promote efficient and scalable electrochemical H2O2 production. While these catalysts demonstrate promising activity and selectivity, improving their long-term stability remains a key challenge. This study aims to understand these structure–activity relationships and develop more robust catalyst designs for practical and scalable applications.
Further reading materials:
J. Mater. Chem. A, 13(44) 38290 - 38300 (2025).
Small Struct., 2 2000051 (2021).
Binghamton University
· Chemistry
Department · 25 Murray Hill Road · Vestal, NY 13850
Materials on this website may only be browsed and saved for personal use.
Please do not reproduce and distribute commercially without permission from
the publisher or website owner.
 Meeting Schedule
Meeting Schedule Weekly Picks
Weekly Picks Research Training
Research Training Shared Resources
Shared Resources Chemical Inventory
Chemical Inventory Presentation Archive
Presentation Archive BU Brain
BU Brain Chem Stockroom
Chem Stockroom Internal Site
Internal Site MyBinghamton
MyBinghamton Group Posters
Group Posters Team Updates
Team Updates Joining our Lab
Joining our Lab Campus Map
Campus Map Campus Parking Info
Campus Parking Info|
|
||